Humanized Mice with CDX Tumor Models
Humanized Immune System Mouse Model engrafted with Human CDX
Contact usCancer remains one of the leading causes of death worldwide, driven by complex interactions between tumor cells and the immune system. Understanding these dynamics is essential for the development of effective immunotherapies targeting human-specific pathways.
Our models enables researchers to evaluate not only the efficacy of immunotherapies targeting various immune compartments – including T cells, macrophages, NK cells, and dendritic cells – but also to gain mechanistic insights into how human immune cells contribute to tumor control and regression.
Main characteristics
-
More than 50 Validated Human CDX Models
With over a decade of accumulated expertise and data, TransCure BioServices offers a diverse portfolio of human CDX models in humanized mice. These models have been thoroughly characterized not only for their tumor growth dynamics, but also for the extent (Hot vs Cold) and quality of human immune cell infiltration, including T cells, macrophages, NK cells, and dendritic cells. This dual profiling ensures that each model provides reliable and insightful data for evaluating immunotherapy efficacy and tumor–immune interactions.
-
Solid and Liquid Models
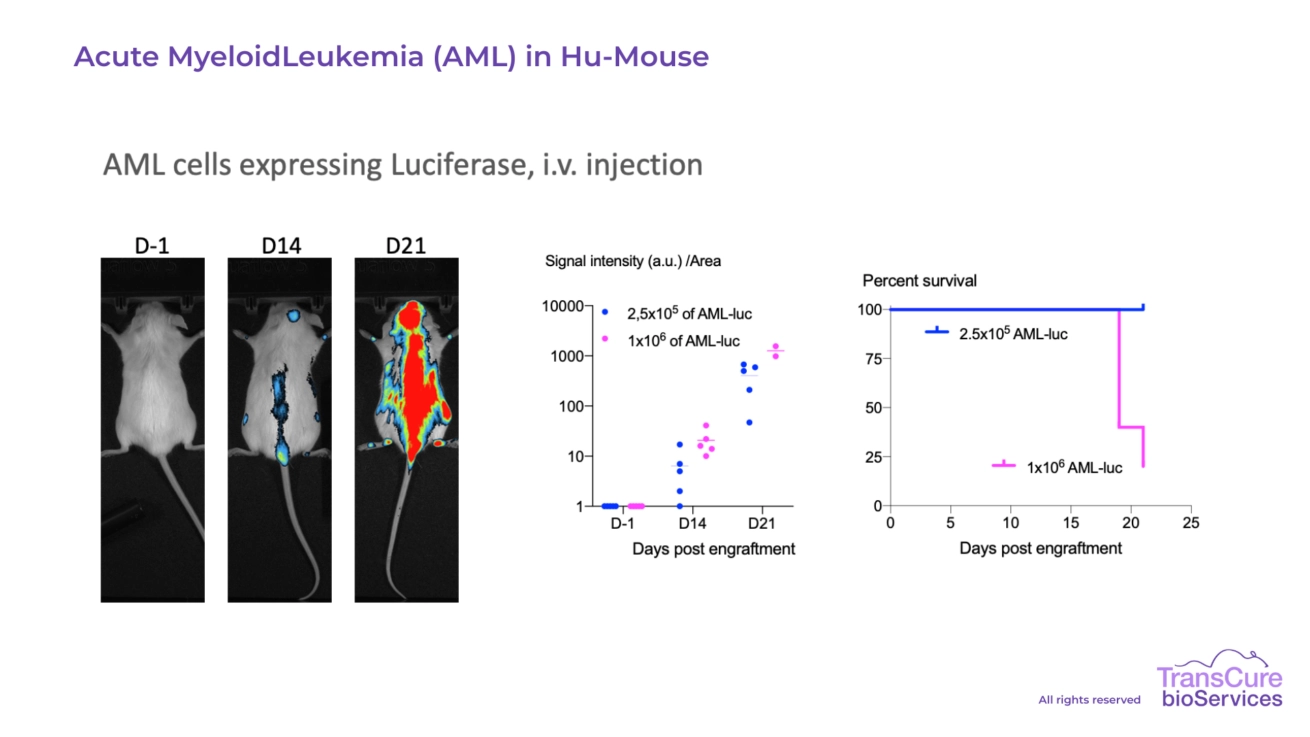
From solid tumor models, where tumor volume is measured using calipers, to liquid tumor models, with tumor burden quantified through non-invasive in vivo imaging, TransCure BioServices offers a range of tailored and validated solutions. Our expertise ensures selection of the most appropriate and predictive model to support your specific clinical application and development goals.
-
Subcutaneous and Orthotopic Model
TransCure BioServices offers both subcutaneous and orthotopic tumor models in CD34+ humanized mice to meet diverse preclinical research needs. Subcutaneous models provide a convenient and reproducible platform for tracking tumor growth and response to therapy using caliper measurements and imaging. Orthotopic models, in contrast, allow tumor implantation in the tissue of origin, offering a more physiologically relevant tumor microenvironment ,metastatic potential, and therapeutic response. Both approaches are supported by our expertise in immune profiling and flexible study design.
-
Tailor Made Models
At TransCure BioServices, we specialize in the development of tailored humanized mouse models to meet the unique requirements of your research program. Whether adapting tumor type, immune profile, route of administration, or specific readouts, our team collaborates closely with you to design and implement customized in vivo studies. Leveraging our extensive expertise in humanized models, we ensure that each project is scientifically sound, flexible, and aligned with your clinical objectives, delivering relevant and translatable data to support your drug development pipeline.
-
TILs Characterization
We have developed robust protocols for tumor dissociation to enable detailed TIL (tumor-infiltrating lymphocyte) profiling by flow cytometry. Our dedicated flow cytometry platform designs custom panels tailored to your specific needs, and our expert team is available to advise on the most appropriate strategies for your study goals. Additionally, we offer TIL analysis by immunohistochemistry (IHC) to provide spatial insights into immune cell localization within the tumor, complementing cellular data with valuable tissue context.
-
PK/PD analysis
Throughout the study, blood samples can be collected at multiple time points and used for PK/PD analysis. We offer in-house capabilities for flow cytometry and CBA-based cytokine profiling, providing real-time insights into drug activity and immune responses. Alternatively, we can process and store plasma or serum and ship samples back to your laboratory for further analysis, offering maximum flexibility to suit your workflow and analytical preferences.
-
Treatment Modalities
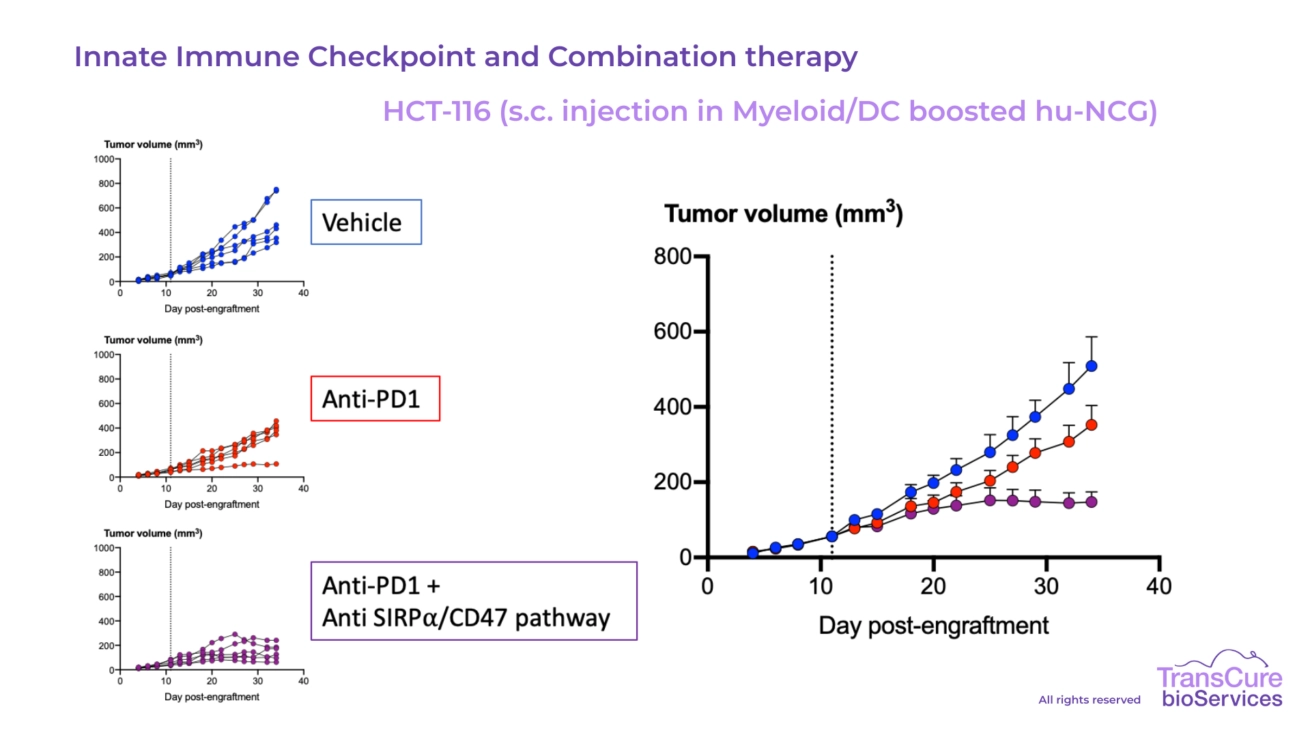
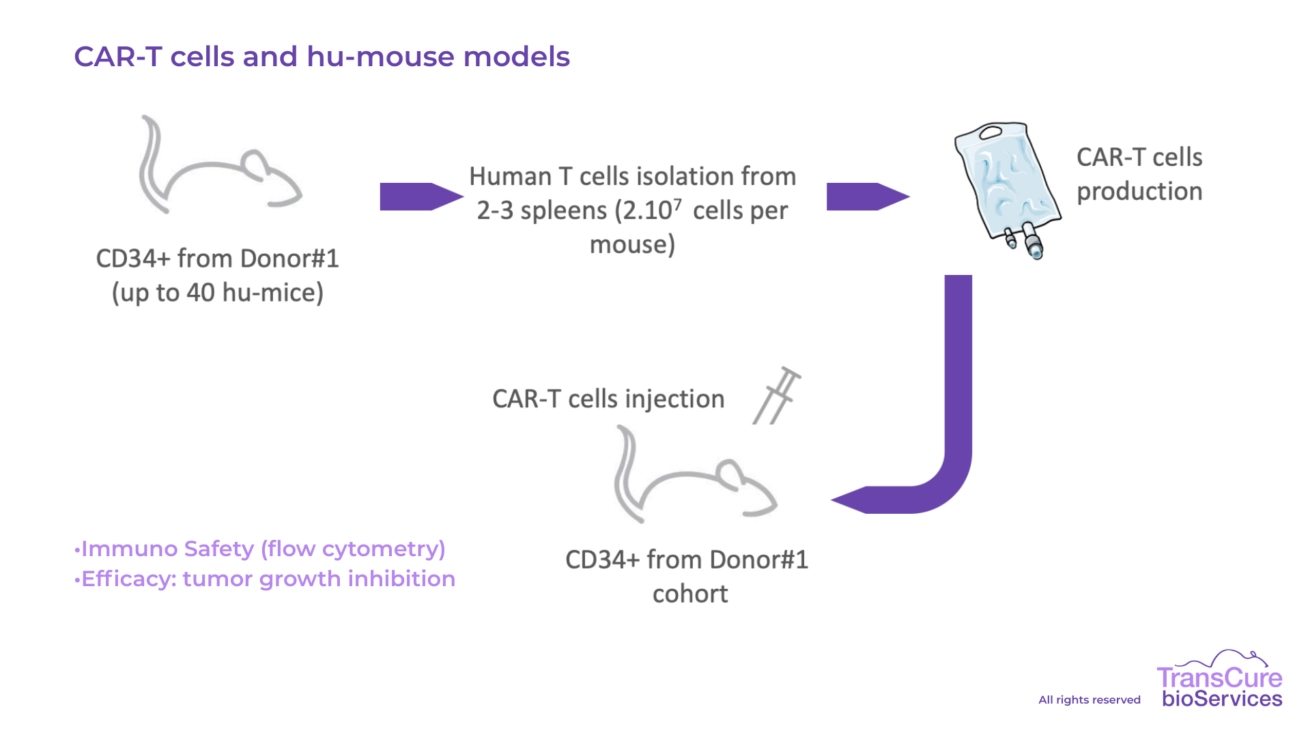
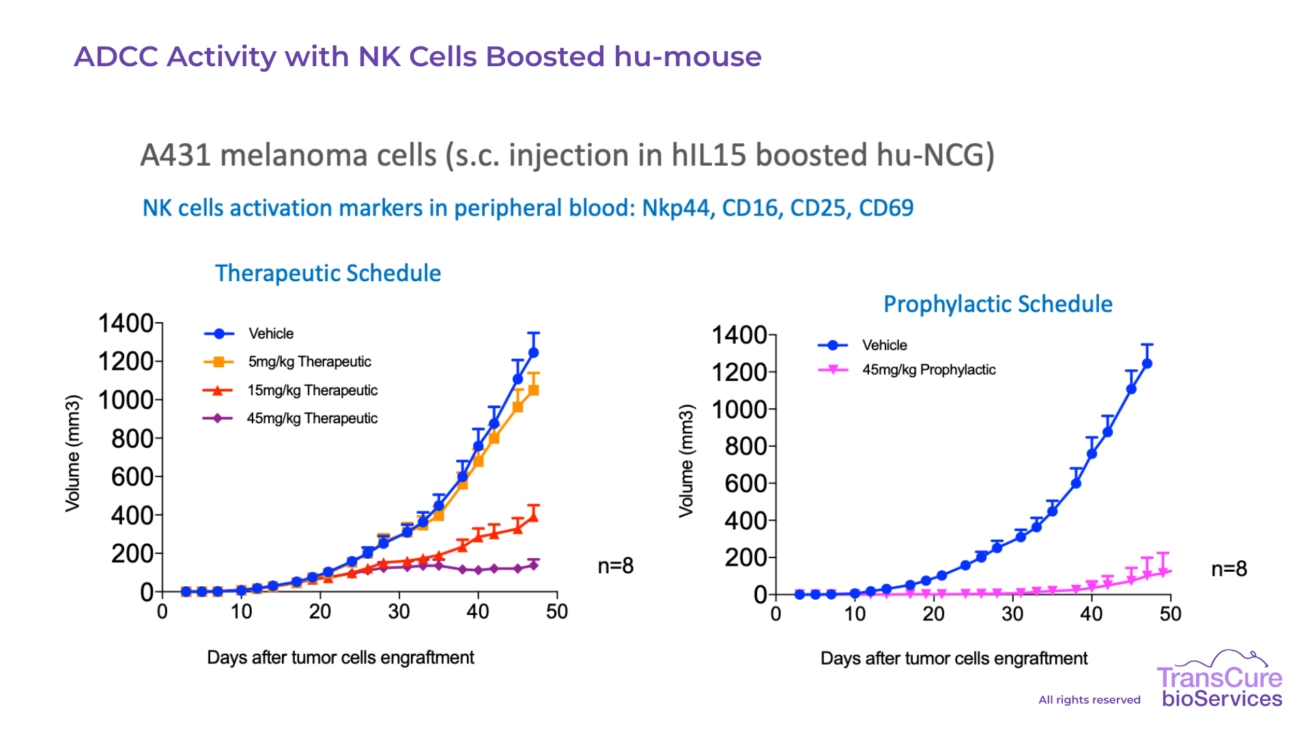
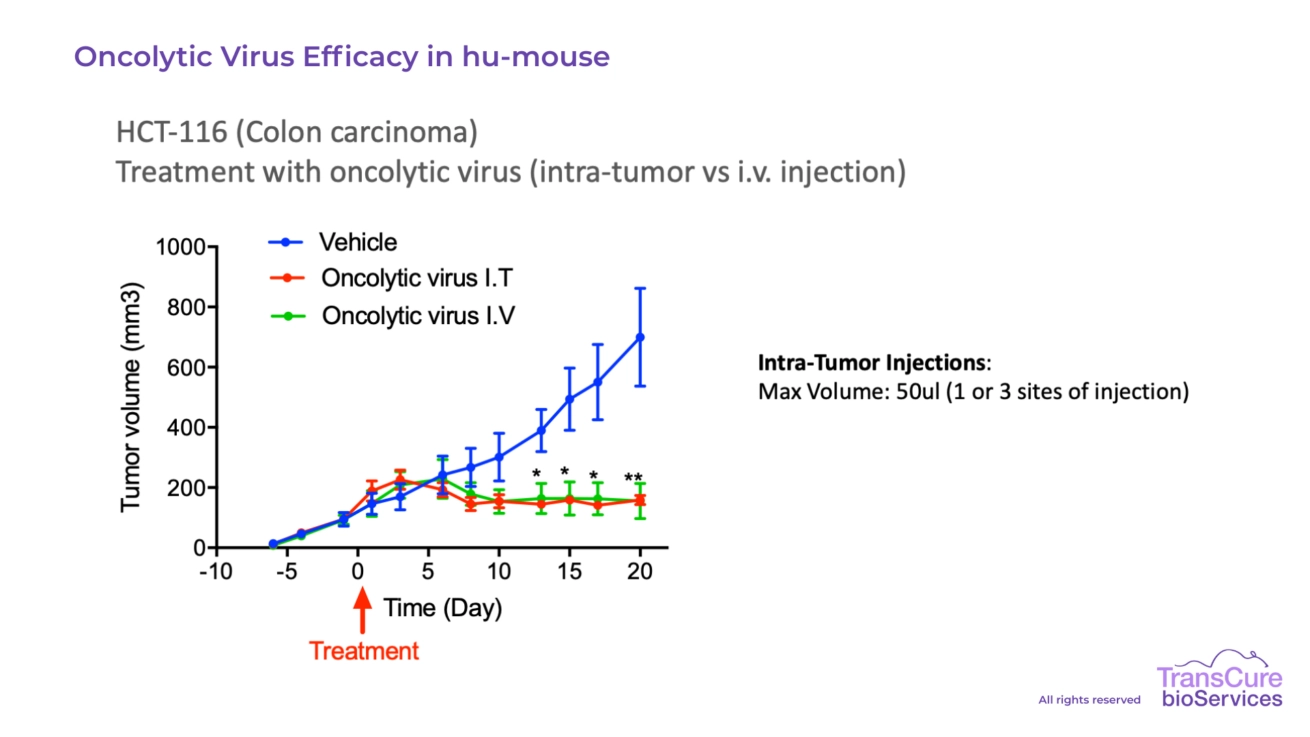
Our CDX models engrafted in CD34 humanized mice are particularly well-suited for testing immune checkpoint inhibitors, T cell engagers, bi-specific antibodies, cellular therapies, ADC, ADCC, Oncolytic virus and cytokine-based treatments, offering high translational value for preclinical oncology research.
Applications
The right mouse model for your research experimentation
-
CD34+ Humanized Immune System Mouse Model
In this model, mice are reconstituted with a fully functional human immune system and engrafted with human CDX or PDX tumors, providing a highly relevant platform to evaluate the contribution of the human immune system in anti-tumor responses. It is particularly well-suited for testing therapies targeting human-specific molecules that do not cross-react with murine counterparts, ensuring translational relevance and mechanistic insight.
See the mouse model
Contact us to have more information about this mouse model
Contact usKnow everything about the CD34+ humanized mouse
-
How do you generate humanized mice?
Humanized mice are generated by engraftment of CD34+ haematopoietic stem cells from human cord blood in immunodeficient mouse strain after a chemoablation treatment. No irradition is performed to avoid anemia and therefore get more robust and stable humanized mice. -
What type of mouse strain can be humanized?
With more than 12 years of expertise, TransCure can humanize many different mouse strains, all have in common to be highly immunodeficient mouse models: NCG, NOG, NOG-EXL… Our team can guide you on the best model to select based on your mechanism of Action. Please note that humanized NCG are prepared in advance to speed up the process. -
How many animals do you recommend per arm ?
For most of our models, starting with a minimum of 6 mice per group provides statistically robust and reliable data. This group size ensures sufficient power to detect meaningful differences while maintaining study efficiency and ethical standards. -
What is the variability due to cord blood donor?
We observe minimal variability between HSC donors in terms of tumor growth and human immune cell infiltration. However, to account for potential donor-specific differences and enhance the translatability and robustness of your results, we generally recommend using 3-4 different donors distributed across experimental arms for most applications. -
Is HLA matching between the tumor and HSC donor necessary?
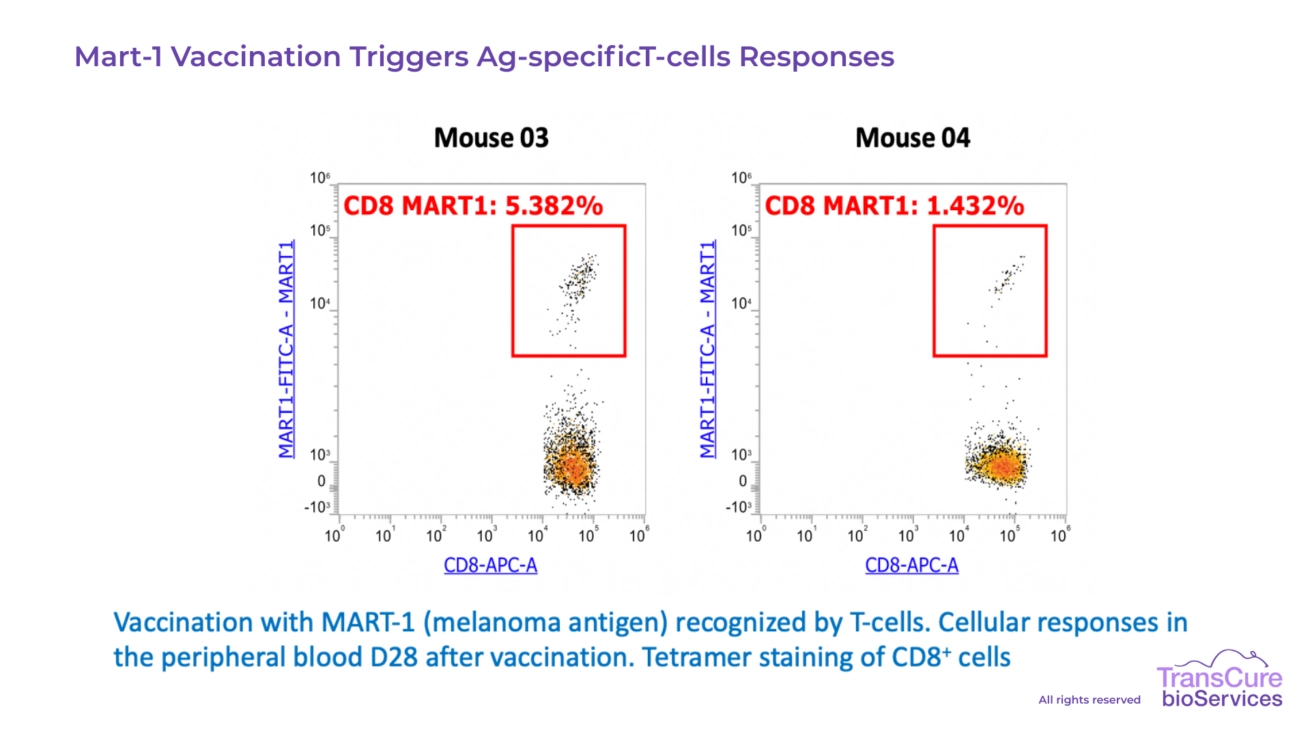
In most cases, HLA matching between the tumor cells and the CD34+ HSC donor is not required for effective engraftment and study outcomes. Our models are designed to tolerate allogeneic settings, allowing for reliable tumor growth and human immune cell function without full HLA compatibility. However, for specific applications-such as studies involving T cell vaccination, or antigen-specific responses, HLA selection and/or matching may enhance the relevance and interpretability of results. We’re happy to discuss the best approach based on your study goals. -
Is it possible to use HSC donors with specific haplotypes?
Yes, absolutely. We can readily select animals humanized with HLA-A*02 by querying our rolling stock, ensuring a rapid and efficient start to your study. Additionally, we offer tailor-made humanization, including pre-screening of HSC donors for specific HLA types, followed by the generation of a dedicated cohort of humanized mice customized to your research needs. This flexibility allows us to align precisely with your experimental design and immunological targets. -
Are the human immune cells from CD34+ humanized mice fully functional?
Yes, the only current limitation is the low level of humoral (antibody) responses typically observed across all CD34+ humanized mouse models. However, this can actually be an advantage in preclinical studies, as it minimizes the risk of anti-drug antibody (ADA) formation, allowing for a clearer assessment of your compound’s pharmacodynamics and efficacy without interference from immune-neutralizing responses.
You have more questions ?
If you have further questions or would like to discuss how our human CDX Tumor models can be tailored to your specific research needs, our scientific team is here to help. We’re committed to providing clear, responsive support and working closely with you to design studies that align with your objectives. Don’t hesitate to reach out - we’re always happy to share our expertise and explore solutions together.
Contact us